Chlorine dioxide gas is one of the most gentle decontaminating agents available. Our process generates a pure chlorine dioxide gas which is used everyday to decontaminate sensitive materials ranging from laboratory scales, microscopes and computers, to complex machinery and entire assembly lines.
Chlorine dioxide is Not Corrosive
Many people are familiar with liquid chlorine dioxide solutions and their corrosive nature. However, these corrosive properties are due to the manner in which these solutions are generated and not the actual chlorine dioxide itself. Many liquid solutions are generated from a typical reaction as follows:
Base + Water + Activator → Acidified sodium chlorite + Chlorous acid + Chlorine dioxide
For most chlorine dioxide liquid solutions, it is these two acidic byproducts, acidified sodium chlorite and chlorous acid that give the solution its low pH (typically around 3) and corrosive properties. However, pure chlorine dioxide of the type generated by ClorDiSys has a non-corrosive neutral pH of 7 when dissolved in water and is gentle on materials. The table below shows common decontaminating agents and their oxidation (corrosion) potential. A higher oxidation potential means that the agent is a stronger oxidizer and thus more corrosive. As can be seen, chlorine dioxide is less corrosive than most of the common decontaminating agents.
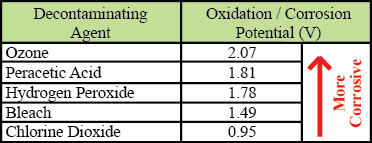
Oxidation Potential
As shown in the table above, chlorine dioxide has a relatively low oxidation (corrosion) potential, 1.9 times lower than hydrogen peroxide. Yet, due to the prevalent use of liquid chlorine dioxide and its corrosive properties, a stigma exists in which chlorine dioxide gas is also considered corrosive. Liquid chlorine dioxide’s corrosive properties stem from its generation chemistry in which multiple acidic components are involved. It is these acids that give the liquid its corrosive qualities, not the chlorine dioxide itself. Our pure chlorine dioxide gas is gentle on materials and should not be considered in the same was as liquid chlorine dioxide products.
Myth: Chlorine dioxide gas forms hydrochloric acid when it contacts water
Fact: Chlorine dioxide gas does not react with water. It stays as a dissolved chlorine dioxide gas in water much like how carbon dioxide is dissolved in carbonated beverages. This unique property allows it to stay effective in solution and kill microorganisms both in the water and on surfaces this water touches. Chlorine dioxide is different than chlorine (which does react with water to form hydrochloric acid) in the same was as carbon dioxide is significantly different than elemental carbon or how diamonds are not at all similar to graphite. Hydrochloric acid cannot and will not be formed in water when using ClorDiSys' pure chlorine dioxide.
Myth: Chlorine dioxide corrodes stainless steel
Fact: Our pure chlorine dioxide does not corrode stainless steel. Stainless steels can be corroded by the acidic byproducts generated along with other manufacturer's chlorine dioxide products.
Myth: Chlorine dioxide gas is not safe on electronics
Fact: Chlorine dioxide gas is safe on most electronics.* We have decontaminated many computers and other electrical devices with no adverse affects. In fact, chlorine dioxide gas was recently chosen to decontaminate the inner chambers of a $3,000,000 Transmission Electron Microscope over hydrogen peroxide vapor for its superior material compatibility as proven through manufacturer testing. Below are test results from an EPA study showing the amount of computer failures after sterilant exposure over a period of 6 months.
The US EPA shows that Hydrogen Peroxide (H2O2) is more corrosive than Chlorine Dioxide (ClO2). Computers were exposed to chlorine dioxide and hydrogen peroxide by the US EPA over the course of 6 months. Chlorine dioxide had the lowest amount of failures. Computers were tested on and off.
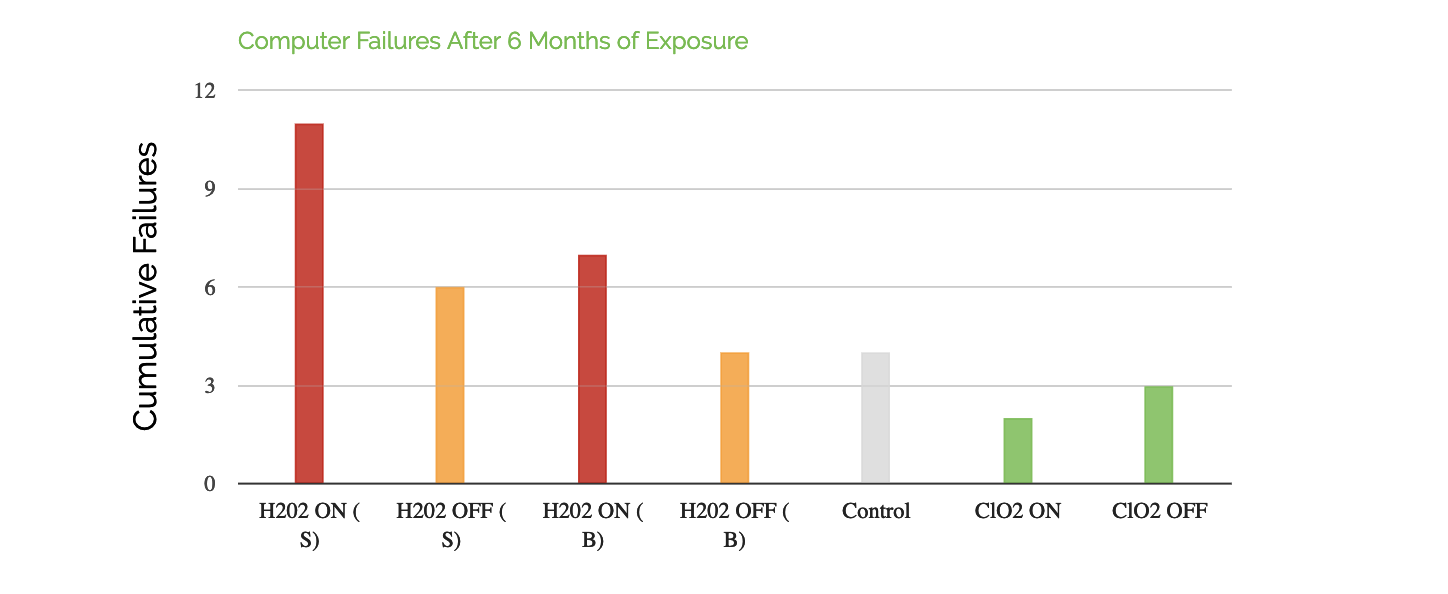
Ref. Snyder, Emily, Indoor and Outdoor Decontamination Presentation at EPA Region 9 / ORD
Homeland Security Research Workshop, July 14, 2011 San Francisco, CA.
*Over time, oxidizers will oxidize; ferrous metals and other sensitive materials may be affected. This goes for all oxidizers, including both chlorine dioxide and hydrogen peroxide. As shown in the chart at the top of this page however, chlorine dioxide has a lower oxidation potential then other common agents.